The Secret Ingredient Is Salt
A collaborative effort reveals previously unknown information about how tiny building blocks come together.

The upgraded X-ray Photon Correlation Spectroscopy (XPCS) beamline at 8-ID of the Advanced Photon Source.
Any great cook will tell you that salt is a critical building block of a strong dish. Turns out, that’s true in the world of nanomaterials as well.
New research out of UT Austin’s Center for Dynamics and Control of Materials, in partnership with national labs, reveals how salt can be used to control the formation and aging of nanocrystal gels, simplifying the development of advanced materials with customizable properties.
Nanocrystal gels are a type of soft material made up of tiny particles, or nanocrystals, that link together to form a network. These gels exhibit unique optical, mechanical and dynamic properties, making them ideal for applications such as sensors, flexible electronics and energy-efficient windows.
However, understanding how these gels form and evolve over time has been a challenge for scientists, especially when molecular additives, such as salts, are introduced to modify their behavior.
“Salt is a common additive in many chemical processes, but its potential to control the gelation of nanocrystals has not been explored,” said Eric Anslyn, a professor in the College of Natural Sciences’ Department of Chemistry and one of the leaders of the study published in Scientific Advances. “Our research shows that salt not only speeds up the gelation process but also reveals a universal pathway that governs how these gels form and age.”
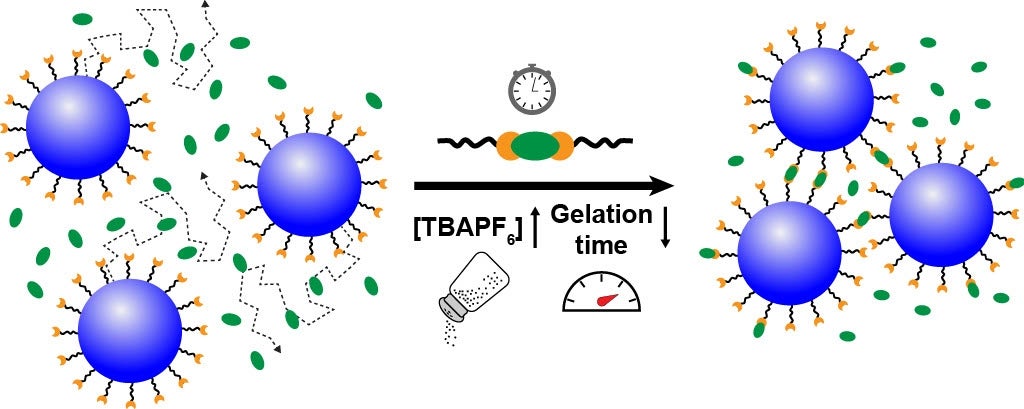
The researchers found that increasing the salt concentration accelerated gelation, enabling the nanocrystals to form networks more quickly. And it did so without altering the gel’s structure.
Salt reduced the electrostatic repulsion between nanocrystals, allowing them to come closer together and form bonds more easily. And it increased the rate of the chemical reaction that links the nanocrystals, further speeding up the gelation process.
“We expected that adding more salt might change the final structure of the gel, but instead, it only affected the speed at which the gel formed,” said Delia Milliron, a professor in the University of Michigan’s Department of Chemical Engineering and former chair at UT’s McKetta Department of Chemical Engineering when the research was conducted. “That means we can predict how these gels will evolve over time and use that knowledge to design materials with specific properties.”
This project was the first to publish using an upgraded X-ray Photon Correlation Spectroscopy (XPCS) beamline at 8-ID of the Advanced Photon Source (APS), a U.S. Department of Energy (DOE) Office of Science user facility at DOE’s Argonne National Laboratory. This is the first research paper published using the upgraded machine, which increased its brightness by up to 500 times.
“The coherence from the upgraded APS combined with Argonne’s leadership computing enabled this class of experiments where the users were able to sample a broad parameter space, which helped the team understand the connection between gelation and the physics that govern the underlying process,” said Argonne scientist and group leader Suresh Narayanan. “This is a great example for future AI-enabled autonomous discovery where users can tune the chemistry of gelation in an optimal manner instead of having to explore the entire parameter space.”
The upgraded APS and the ultrafast detector at the 8-ID beamline allows scientists to routinely perform XPCS measurements with 20-microsecond time resolution with high fidelity within the blink of an eye, a measurement that previously would have taken an hour. Researchers can use X-ray beams to probe materials and observe them at the atomic scale without damaging them. Scattering the X-ray beam off a sample over a designated time period allows scientists to examine the way that sample relaxes, diffuses or reorganizes over that period.
“This brings crucial new insight because it allows us to probe the exact temporal and spatial scales that directly correspond to the dynamics at the size of the nanocrystals,” said Argonne scientist Qingteng Zhang.
While the study has provided valuable insights into the role of salt in nanocrystal gelation, the researchers acknowledge that there is still much to learn. For example, the current models used to describe the gelation process do not account for certain complexities, such as charge regulation and many-body interactions. Future research will aim to incorporate these factors into more comprehensive models.
The researchers also plan to explore other ways to control gelation, including adjusting pH, solvent properties and the size and charge density of the nanocrystals. These efforts could lead to the development of reconfigurable, programmable materials with applications spanning energy storage to biomedicine.
The study was supported primarily by the National Science Foundation (NSF), with additional support from the Welch Foundation, and the U.S. Department of Energy (DOE). The full team includes: William D. Brackett, Dingwen Qian, Tsung-Lun Lee, Jiho Kang, Jinny Choi , Allison M. Green of the McKetta Department of Chemical Engineering; Charles K. Ofosu, Diana L. Conrad, Anna Bessmertnaya, Jessica D. Oberlander of the Department of Chemistry; Thomas Truskett of the University of Michigan’s Department of Chemical Engineering; Suresh Narayanan, Qingteng Zhang and Miaoqi Chu of Argonne National Lab’s Advanced Photo Source; Felix Lehmkühler of the Hamburg Centre for Ultrafast Imaging in Germany; and Andrei Fluerasu of Brookhaven National Laboratory.
